Tissue Culture for Cannabis
List of contents
Micropropagation is one of the most widespread uses of in vitro plant cultivation. These techniques and tissue culture methods are used to multiply genetically identical plants (known as clones) on a large scale, in reduced spaces and short periods of time, combining some of the advantages of both systems of plant reproduction.
This method of in vitro plant propagation is one of the plant biotechnology cultivation techniques that has shown a huge rise in popularity in recent years, as it allows growers to obtain thousands of identical plants from very small sections of a mother plant, called explants. The success of any micropropagation will depend on the ability to acclimatise high-quality plants from in vitro conditions to ex vitro conditions, thus producing new plants from these explants.
In this article, we'd like to take a closer look at the fascinating world of tissue culture, outlining the possible applications that this technique offers to cannabis cultivation which, as you'll see, are quite a few!

Introduction to the history of tissue culture
The story of plant tissue culture began in 1902 when the Austrian botanist Gottlieb Haberlandt (1854-1945) standardised plant cell and tissue culture techniques with the concept of cell totipotency (the idea that all plant cells have the ability to regenerate into whole plants). For this reason, Haberlandt is today considered the father of tissue culture. In 1930, based on these discoveries, plant hormones were introduced, offering the possibility to guide the morphogenetic programming of cells and thus promoting organised and disorganised cell growth.
Then, in 1955, Miller discovered cytokinins, which are one of the most important types of hormones in cell differentiation and division and are fundamental in the process of organogenesis. Later on, in 1960, Kanta successfully achieved vegetative propagation of virus-free orchids by culturing apical meristems. A few years later, in 1962, Murashige and Skoog developed the MS culture medium, which is currently the most common and widely used for in vitro culture of plant tissues.
The term micropropagation was first defined in 1986 and today it has become an important technology in the selection and crossing of plants as well as the control of plant diseases.
Stages of plant micropropagation
Micropropagation starts with the objective of cloning plants that have the desired characteristics, quickly and in massive numbers. The process has several very different stages, with perfectly defined objectives for each of them. It should be noted that all the plant material that will be cultivated in vitro will be genotype-dependent, which means that each action or protocol that is applied to the culture will have a different response in each plant genotype.
Within the micropropagation process itself, there are several different phases or stages that together make up a complete micropropagation cycle. This method can be applied to different plant species, with the ability to simplify the process or add further stages according to the characteristics of each plant, but in general terms, these are the most common steps of the in vitro reproduction process of plants.
A clear example is that of woodland species, which require an extra stage called hardening, which occurs before being planted out in the field and after ex vitro acclimatization.

Selection & Preparation of the mother plant
The proper choice of starting material for in vitro culture - and more specifically for micropropagation - does not make sense if the plant or individual to be cloned lacks certain determining and contrasting qualities and genetic traits, at the very least enough to allow a program of mass multiplication of the chosen individual.
On many occasions, the genotype used as starting material is determined by the results of selection programs, where the best plants are chosen as mothers for their subsequent in vitro reproduction.
Selecting cannabis mother plants
Choosing a good mother plant is not easy, especially if you have very specific goals in mind. To make it simpler, this article covers the main factors that are usually considered when selecting a mother plant; which, as you will see, can vary a great deal depending on the type of grower.
Sterilising the plant material
This stage consists of the sterilisation of the explants by means of multiple disinfections and several treatments. Once the mother plants have been selected, it is necessary to disinfect the surfaces of all the plant material in order to prevent unwanted microorganisms such as bacteria and fungi from developing in the culture medium.
The choice and concentration of the disinfectant agents and the disinfection time employed are mainly determined by the characteristics of the explant; In practice, it basically consists of following the trial and error method until a good protocol is found for each material, always with the aim of eliminating external contaminants. At this stage, it is about finding a balance between optimising the disinfection process while causing the least possible damage to the plant material itself.
Placing the material in vitro
The objective of this phase is to introduce the explants into the culture medium via the chosen route. Based on this, we can talk about:
- Meristem culture
- Bud culture
- Protoplast culture
- Anther culture
- Embryo culture
This work is always carried out inside a laminar flow cabinet to avoid unwanted contamination. Once the plant material has been disinfected, it is placed in a sterile culture medium and left to begin to sprout, which can take one or two weeks.
Multiplication through subcultures
This stage or phase consists of obtaining the maximum amount of plant mass from which to take the new explants later on. The objective of this phase is to induce the multiplication of enough explants to regenerate the number of plants required. In this process, culture media, growth regulators, cytokines and gibberellic acids are all used, and it is usually referred to as the germination or regeneration process of new plant tissues.
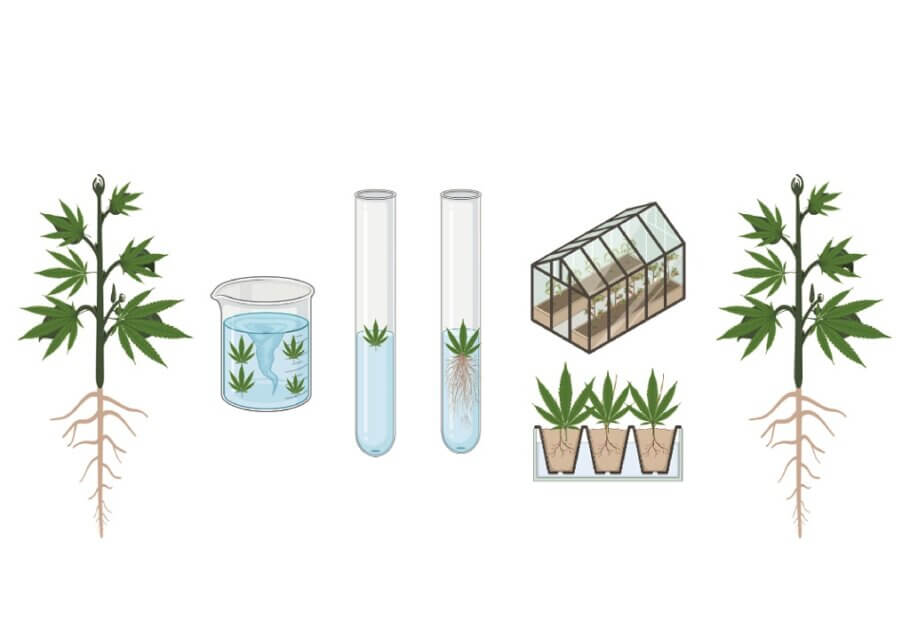
Rooting (in vitro vs ex vitro)
As its name indicates, the purpose of this stage is to stimulate and promote the formation of roots in vitro plants, a process that can be carried out in vitro or ex vitro. The decision whether to root in vitro or ex vitro will depend greatly on the type of material being cultured and its ease of rooting.
Acclimatisation ex vitro
Acclimatisation allows plants to adapt to life in natural conditions. If rooting has been carried out ex vitro, acclimatisation will take place at the same time.

On the other hand, if rooting is carried out in vitro, then the plants must then be acclimatised to external conditions. Because plants that have been grown under test tube conditions usually have non-functional stomata, all plants must be adapted to acclimatisation periods in which the relative humidity of the environment is gradually lowered until we have plants with functional roots.
In this phase, we also gradually increase the intensity of the lighting system being used. Needless to say, the success of micropropagation consists largely in the number of plants that we manage to acclimatise!
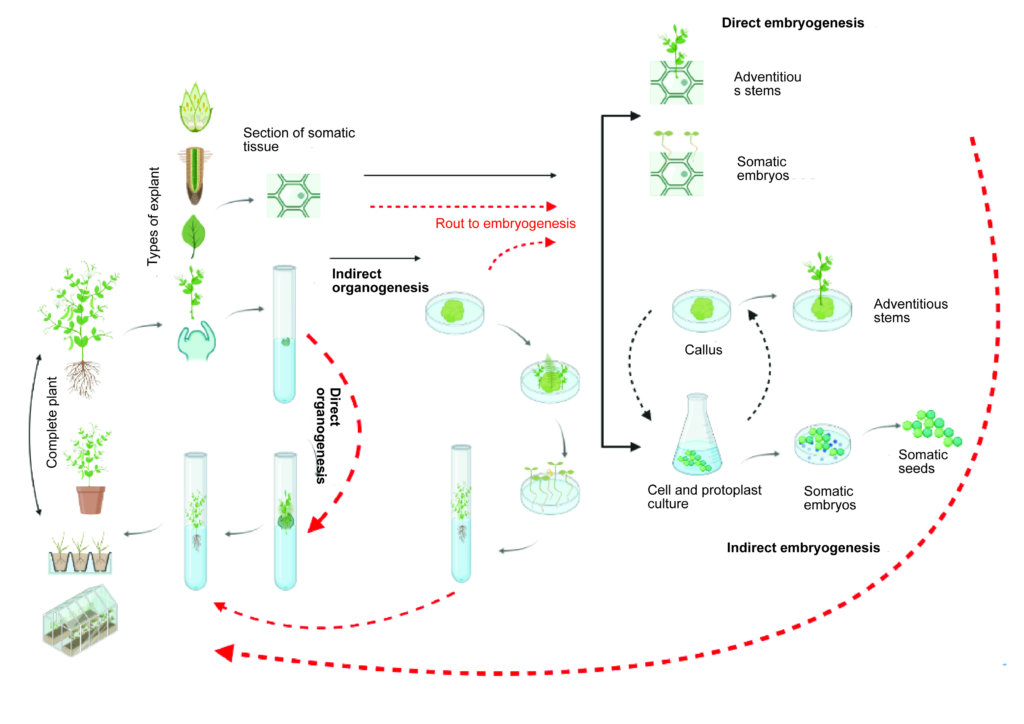
Types of micropropagation
Micropropagation is based on multiplying explants en masse under controlled and sterile conditions. In in vitro culture, we can use several types of explants, each with a function or route to follow. The most used explants for in vitro propagation processes are the vegetative buds of plants.
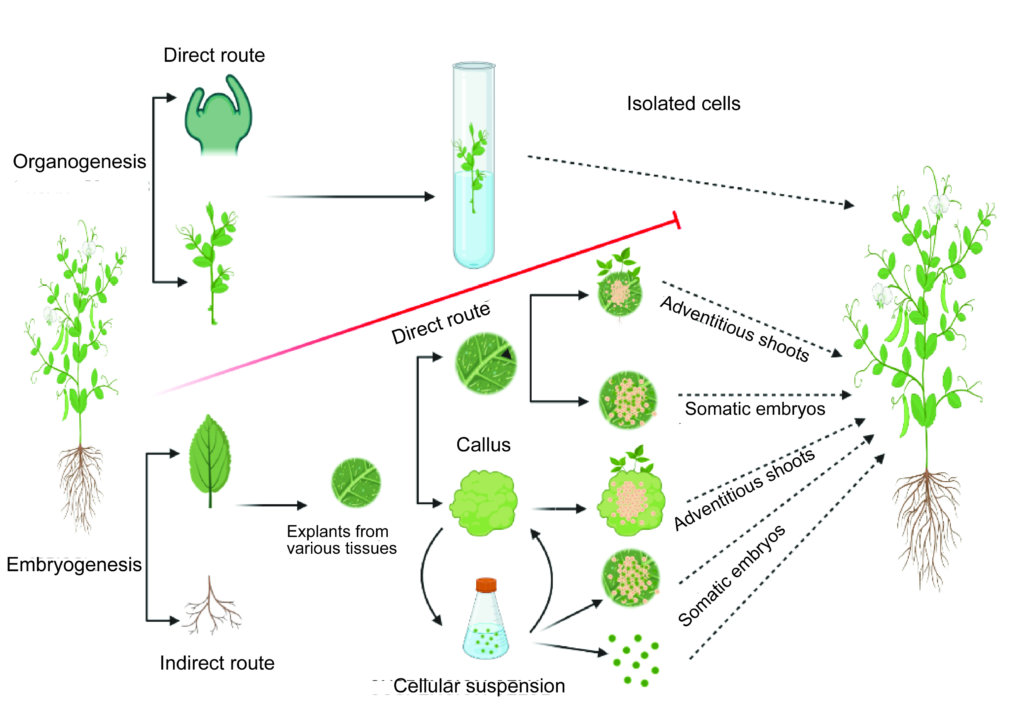
There are two main routes when it comes to the micropropagation of plants in vitro, organogenesis and embryogenesis:
What is organogenesis?
Organogenesis is a process that consists of obtaining stems, roots or flowers from a bud or shoot. It is worth noting that this method requires several events depending on the type of tissues to be regenerated, whether they are shoots or roots.
Organogenesis can be direct (from the explant) or indirect (from the callus):
- Direct organogenesis: A morphogenetic response by which organs are directly formed in order to regenerate whole plants.
- Indirect organogenesis: A morphogenetic response by which the formation of organs is achieved from callus culture. Micropropagation via callus has the possibility of somaclonal variation, leading to genetic instability, a loss of uniformity and other undesirable results, and therefore is not so valued in clonal propagation.
What is embryogenesis?
Embryogenesis is a process that seeks the formation of somatic embryos, and is also called asexual or adventitious embryogenesis. The resulting embryos germinate, giving rise to a stem and a radicle simultaneously (in a single event), just like a cannabis seed does when it germinates. The success of somatic embryogenesis depends on the variety introduced and the supply of growth regulators in the culture medium.
Just like organogenesis, embryogenesis can be direct or indirect:
- Direct embryogenesis: A biological process by which somatic cells develop embryos. Somatic embryogenesis is direct when stems or embryos are produced directly from the starting tissues.
- Indirectembryogenesis: This implies the need for induction so that the cells of the explants used are disordered and form a callus before following the embryogenic pathway of common development.
Future prospects of in vitro culture
One of the main challenges in micropropagation today is the standardisation and up-scaling of the processes or protocols that must be followed so that they can reach a point of profitability of the business, thus creating a niche in the market for price, time, and quality of the product.
Automisation is another of the great factors that could influence the final price of an in vitro plant, as many of the techniques or routes used in micropropagation require many hours of human labor. In this way, if the process - or part of it - could be automated or robotised, the final cost of a tissue cultured plant would greatly decrease.
Bioreactors are another exciting development in tissue culture research. There are many types and forms of bioreactors but their principal function is to optimise the multiplication of the plants produced in this type of culture. The main purpose is to automate and lower production expenses, reducing labour costs and achieving higher multiplication rates than in other systems. Bioreactors are also characterised by using liquid culture media in which the addition of gelling agents such as agar is not required.

Finally, biofactories are large-scale in vitro plant propagation laboratories. These tissue culture factories are characterised by having very limited sowing processes, with fully structured plant propagation steps and routes. The main purpose of a biofactory is to have highly optimised and standardised settings and protocols with a clear objective, which is none other than the maximum production of plants in the minimum time, space and cost. To date, biofactories have successfully processed forest, fruit, aromatic and ornamental species.
That's it for this introduction to the exciting world of in vitro plant propagation. The truth is that the range of possibilities that these methods open up, especially in large-scale cultivation, is impressive. Please feel free to leave any questions, or comments about your own experiences and, as always, we'll be happy to answer you.
Happy (micro) growing!

































































