MARIJUANA AND MEDICINE: CESAMET®, MARINOL®, SATIVEX®
List of contents

We know that cannabis has been used as a natural remedy for thousands of years. Today, scientific research corroborates this knowledge, which is continuously extending.
Cannabis as a medicine is considered as a palliative for symptoms of diseases such as fibromyalgia, neurological diseases like multiple sclerosis, glaucoma, effects of cancer treatments, to treat certain types of chronic pain and for some autoimmune diseases, etc..
Pharmaceuticals have been trying to standardise the active principles of the cannabis plant for years, both in its formulation and composition, as well as in its dosage. These paths have led to the marketing of three drugs: Cesamet ® (nabilone), Marinol ® (dronabinol) and Sativex ®
CESAMET® (nabilone)
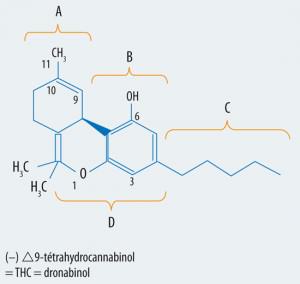
Cesamet, name which recieves the Nabilone on the market, is a synthetic analogue of THC, which is not the same as synthetic cannabis, i.e.
it presents the same structure to be identified by the recipients but with some changes in its composition.
Nabilone was introduced in the mid-80's in the United States and was then removed at the end of that decade. It was used as an antiemetic, to alleviate symptoms such as nausea and the consequent vomits from anti-cancer treatments.
Valeant Pharmaceuticals International, based in California, bought the patent in 2004 and brought it on to the market under the name Cesamet ® during the middle of the last decade. It was also sold in Canada, Australia, United Kingdom and Ireland, extending then to other countries, but not as a drug of first choice. Cesamet ® is available in a bottle of 20 capsules with 1 mg or 1.5mg of nabilone.
MARINOL® (dronabinol)
There is another synthetic equivalent of THC called dronabinol; it has the appearance of a brown oil and is administered in glycerine capsules of 2.5, 5 and 10mg.
It is marketed under the name Marinol ® in the United States, South Africa, Australia and New Zealand. It has the same particulars as Cesamet ® and is also used to alleviate the symptoms of anorexia / cachexia syndrome in patients with HIV or terminal cancer.
We must say that synthetic cannabinoids have shown side effects when not used with their THC antagonists, such as CBD and others - which serves to counteract some effects - so they decided to make extractions directly from plant being in order to mantain the range of cannabinoids as much as possible.
SATIVEX®
Unlike these two drugs, Sativex ® is not a synthetic cannabinoid, but an extraction of a selected phenotype - a clone - with a

number of known, standardised and homogeneous cannaboid, which basically contains delta-9-tetrahydrocannabinol (THC ) and cannabidiol (CBD).
A dose of 0.1 ml contains 2.7 mg of THC, 2.6 mg of CBD and 5% of other cannabis derivates. It is administered sublingually by spray-application, and it is sold in bottles of 5, 5 ml with 51 applications. It is aimed, for the moment, at patients with multiple sclerosis and to treat pain in cancer patients. It was patented by the British laboratory GW Pharmaceuticals at the beginning of the last decade and marketed by Bayer and Laboratorios Almirall; this last laboratory is carrying out a pilot-test in Catalonia.
We can see that this kind of treatments with synthetic cannabis derivates have a limited use within the medical cannabis scene. Many therapeutic consumers maintain a firm stance on using only the marijuana plant.
For them, cannabis remains as the best way to treat their ailments. Their attitude is the spearhead against the greedy interests of the big pharmaceuticals in taking advantage of the emerging world of medical cannabis and its great economic potential.